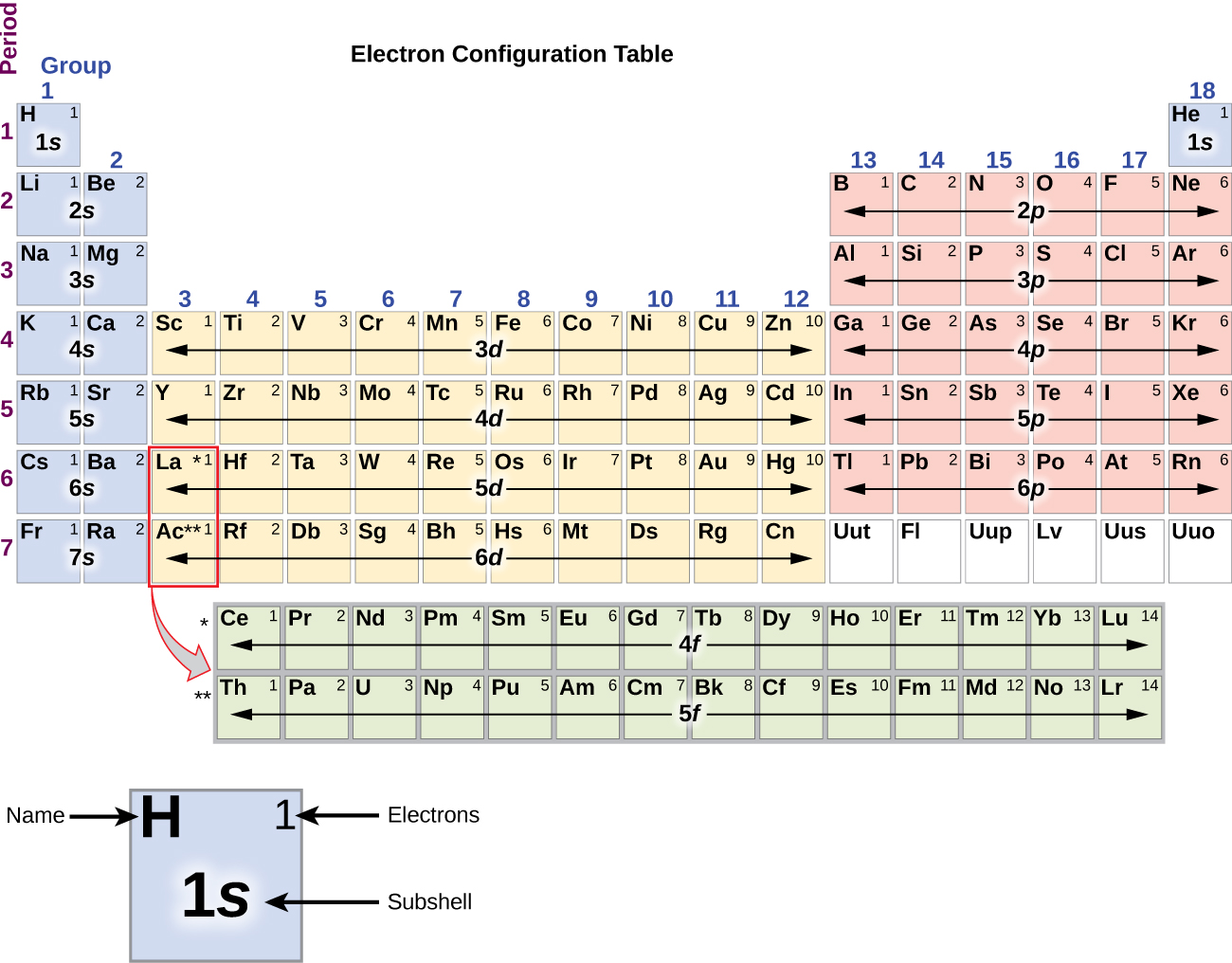
/ColorPeriodicTableEC-58b5c7fa3df78cdcd8bbb56f.png)
The electrons’ distribution into the atomic orbitals is called as a. This Jekyll and Hyde behavior of 3 d electrons makes life more complicated (and often far more interesting) for chemists who study the transition elements. Chemistry Multiple Choice Questions on Electronic Configurations of Elements and the Periodic Table.
#Electron configuration periodic table full
In these cases, a completely full or half full d sub-level is more stable than. Although the 3 d electron clouds do not extend farther from the nucleus than 3 s and 3 p (and hence do not constitute another shell as the 4 s electrons do), they are thoroughly shielded from the nuclear charge and thus often act as valence electrons. There are two main exceptions to electron configuration: chromium and copper. In some cases electrons in the d subshells act as valence electrons, while in other cases they do not. The chemistry of the transition metals is more complicated and a wider variety of formulas for transition-metal compounds is possible because of this variable valence. For example, in some compounds V (vanadium) has a valence of 2 (VO, VCl 2) in others it has a valence of 3 (V 2O 3, VCl 3), in still others it has a valence of 4 (VO 2, VCl 4), and in at least one case (V 2O 5) it has a valence of 5. The valence of 2 corresponds with the two 4s valence electrons.Įach of the transition metals also exhibits other valences where one or more of the 3 d electrons are also involved. This article provides you with an electronic configuration chart for all these elements. Each element has a unique atomic structure that is influenced by its electronic configuration, which is the distribution of electrons across different orbitals of an atom. There are 118 elements in the periodic table. This can be seen in the table found at the top of this page. Electron Configuration Chart for All Elements in the Periodic Table. Notice, for example, that except for Sc, all of the transition metals form chlorides, MCl2, where the metal has a valence of 2 examples are TiCl2, VCl2, CrCl2, and so on. Hence there is a slow but steady transition in properties from one transition element to another. Adding one more 3 d electron has considerably less effect on their chemical properties than adding one more 3 s or 3 p electron did in the case of the representative elements. The 3 d electrons are “buried” under the surfaces of the atoms of the transition metals. 1 Electron Configuration Battleship gameplay is similar to the board game, Battleship, popularized by Milton Bradley. Thus the 4 s electrons are often more significant than the 3 d in determining valence and the formulas of compounds. Similarity of valence shell electron configuration implies that we can determine the electron configuration of an atom solely by its position on the periodic. Figure 1: Periodic Table with valence electron configurations. \( \newcommand\) ) interacts with another atom, it is the 4 s electrons extending farthest from the nucleus which first contact the other atom. The symbols used for writing the electron configuration start with the shell number (n) followed by the type of orbital and finally the superscript indicates.
